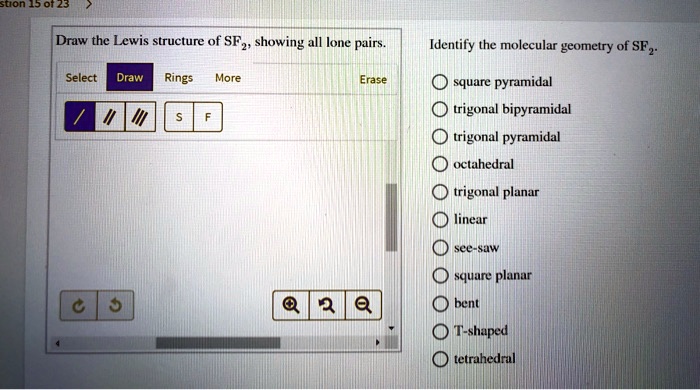
The sulfur atom has two bonding pairs of electrons and two nonbonding pairs of electrons that represent the VSEPR notion of AX2E2, which corresponds to an angular/non-linear or bent molecular geometry. The lower plot shows the residuals of the linear regression, indicating that the sample does not show aggregation or repulsive effects. A quick explanation of the molecular geometry of ClO2 - (Chlorite ion) including a description of the ClO2 - bond angles.Looking at the ClO2- Lewis structure. The molecular geometry of the molecule depends on the Lewis structure and the arrangement of valence electrons in the structure. The concentration of the sample was 5 mg ml −1. ( e) Guinier plot of hsRIG-I SF2 SAXS data with linear regression (for points shown in red) used for determination of radius of gyration ( R g = 3.37 ± 0.01 nm). ( d) Cartoon representation of the crystallographic dimer with one monomer shown as in ( a) and the second one in light blue. Kowalinski et al., 2011 ) RIG-I SF2 crystal structures. Civril et al., 2011 ) and duck (orange, PDB entry 4a2p ( c) Overlay of human (blue, this study), mouse (red, PDB entry 3tbk Identify the electron-group geometry, molecular structure. The well coordinated sulfate ion occupies the space of the β-phosphate of the nucleotide-bound state. Check Your Learning Build a more complex molecule in the simulator. Sulfur difluoride, SF2, is an AX2E2 species, and it has an angle of 98 degrees.

In NO2+, there is no repulsion taking place between the two O atoms and the lone electron on the central atom. , shown as lines Kowalinski et al., 2011 ) and the corresponding region of the structure reported in this study (shown as sticks). NO2 is a bent molecule however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. ( b) Superposition of the ATP-binding site of the RIG-SF2 ADP-AlF 3-bound structure (PDB entry 4a36

Subdomains are represented in different colours: Hel1, red Hel2, brown Hel2i, blue bridging domain, orange. BASSMIDIFONTLINDECVOL: Use linear decay and release phases in volume envelopes. The attack phase is always linear in SFZ envelopes. ( a) Crystal structure of human RIG-I SF2. Use a linear attack phase in SF2 modulation (pitch/filter) envelopes.
